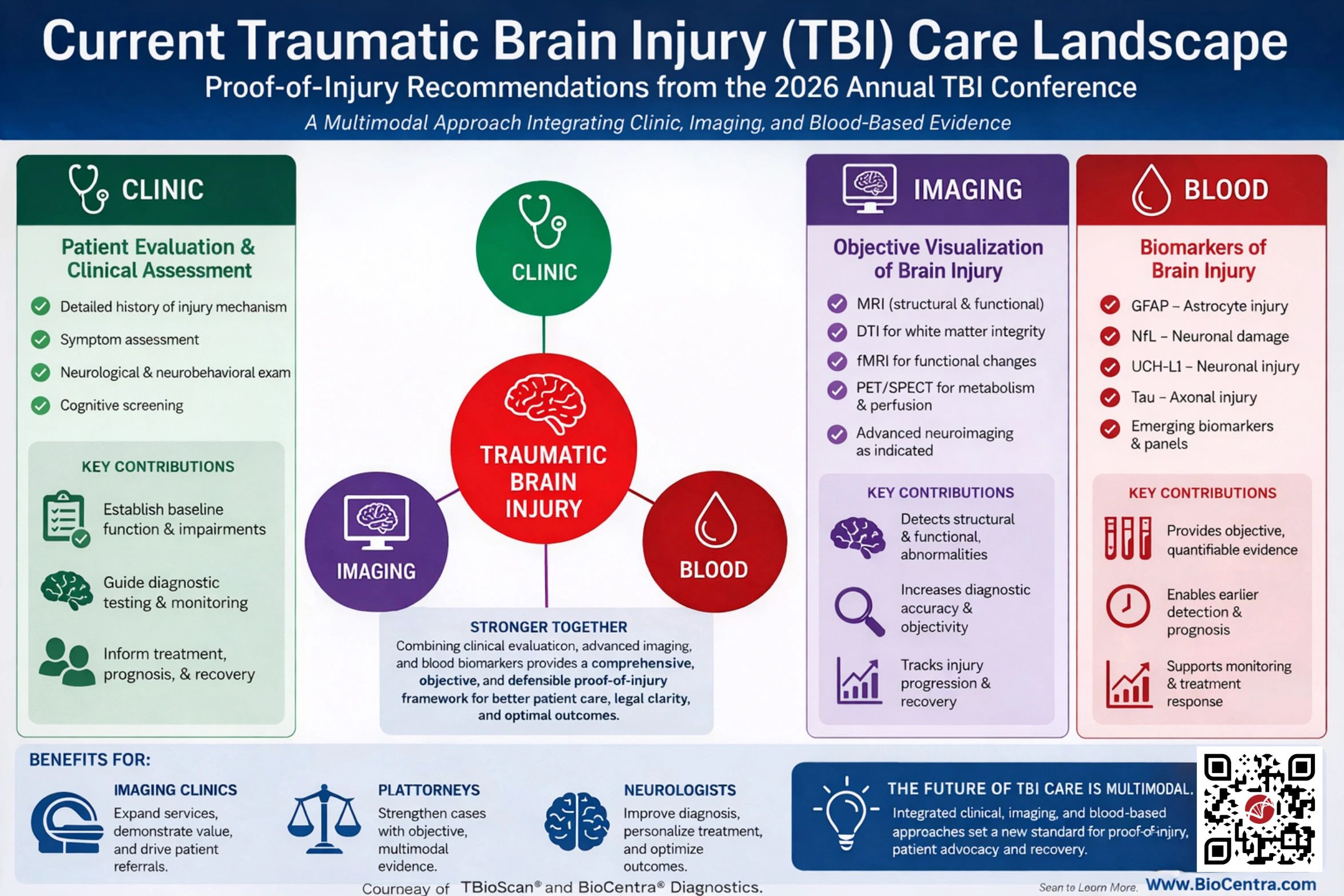
Our TBioScan® technology is redefining the way NEUROLOGISTS look at mild TBI cases.
Redefining Standards in Traumatic Brain Injury (TBI), Alzheimer’s Disease and Cardiovascular Diagnostics
Our TBioScan® technology is redefining the way NEUROLOGISTS look at mild TBI cases.

Redefining Standards in Traumatic Brain Injury (TBI), Alzheimer’s Disease and Cardiovascular Diagnostics
At BioCentra®, we are committed to providing trusted services and to ensure accurate, reliable and timely results to all our clients.
We love our customers, so feel free to visit during normal business hours.
Open today | 09:00 am – 05:00 pm |
Get 10% off your first purchase when you sign up for our newsletter!

Order a NextGen Screening Test for Dementia/ AD, TBI or Cardiovascular Diseases (CVD) Today!